Session Highlights
Summary
- The session aimed to talk about the Wallerian degeneration pathway and the molecular mechanisms identified over the past 20–30 years and how this is relevant to the low value regeneration seen in most peripheral neuropathy patients.
- NMNAT2 basically has an inhibitory hold on SARM1 activation, so if NMNAT2 levels are high in axons, SARM1 doesn’t get activated and there is no axonal degeneration.
- It is this close relationship between NMNAT2 and SARM1 activation that allows broad thinking about inhibiting SARM1 as a common final pathway in many different, predominantly axonal neuropathies.
Types of axon degeneration:1
- Developmental axon pruning: Terminal pruning and large-scale pruning
- Wallerian degeneration: Traumatic or ischemic injury
- Dying back distal axonal degeneration: Metabolic, toxic, genetic injury
Wlds and Wallerian Degeneration2
- WldS mutation spontaneously arose.
- Transected axons survive for long periods of time
- WldS partially protects axons against “dying back” Wallerian-like degeneration
- WldS is a fusion protein of Nmnat1 and a 70-aa fragment of Ube4b
Wallerian degeneration vs distal axon degeneration: Role of NMNAT3,4
- Data from WLDS suggest that axonal NMNAT plays a role in some but not all distal axonal PNs
| WLDS partially protective | WLDS NOT protective |
|---|---|
| Paclitaxel induced neuropathy3 | CMT2D (Gars model)3 |
| Glaucoma models4 | SMA (2 different models)3 |
| Parkinson’s models4 | ALS (SOD1 mutations)3 |
| CMT1B/CMT1A3 | HSP (Plp mutant mouse)4 |
| EAE3 | Prion disease3 |
| STZ model of diabetes3 | |
| Tauopathy4 |
Loss of NMNAT2 induces Wallerian degeneration
- RNAi of NMNAT2 but not NMNAT1 or NMNAT3 induces axon degeneration in SGC neurons3
- Axon degeneration induced by NMNAT2 RNAi is delayed by WLDS5
- NMNAT2 is the most labile isoform but overexpression of NMNAT2 delays axon degeneration4
Mutations in dSarm block Wallerian degeneration5
- Forward genetics in drosophila identified dSarm as a key regulator of axonal degeneration
- Deletion of mouse homologue, SARM1, delays axon degeneration after sciatic nerve transection
Aberrant activation of programmed axon death causes disease6
- Loss of NMNAT2 causes developmental failure of axon growth
- This is rescued by WldS
Aging and axonal NMNAT27
- Brain NMNAT2 expression level correlates with cognitive function in AD autopsies
- NMNAT2 transport decreased with aging
NMNAT2 mutations in human disease8
- Compound heterozygous NMNAT2 variants in 2 brothers with relapsing-remitting neuropathy
- Nmnat2V98M/R232Q mice have behavioral and electrophysiologic features consistent with a motor neuropathy
Neuronal SARM1 is required for Nmnat2V98M/R232Qneuropathy9
- Both Sarm1 KO and dnSarm1 are protective
GoF mutation of Sarm1 in human disease10
- Constitutively active SARM1 variants found in ALS patients induce axon degeneration
- Mice injected intrathecally with a SARM1V184G AAV construct develop motor axon degeneration
Does Sarm1 deletion protect against distal axon degeneration as seen in axonal PN?11
- Protects against vincristine neuropathy
- Outcome measures (IENFD, tail SNAP amplitude and axon degeneration in sural nerve)
Sarm1 KO protects against HFD induced PN12
- HFD model of metabolic neuropathy
- Sarm1 KO mice on HFD do not develop peripheral neuropathy
Sarm1 KO protects against bortezomib induced PN13
- BTZ and VCR activate different pathways that converge on Sarm1
Sarm1 KO protects against STZ induced PN14
- Protects against STZ model of Type 1 diabetes induced neuropathy
Implications:
- The discovery of the NMNAT2–SARM1 axis has fundamentally changed the understanding of axon degeneration, revealing it as a druggable process. This offers a transformative opportunity for developing targeted, mechanism-based therapies for peripheral neuropathies and potentially broader neurodegenerative disorders
Abbreviations
AAV: adeno associated virus, AD: Alzheimer’s disease, ALS: amyotrophic lateral sclerosis, AxPN: axonal peripheral neuropathy, BTZ: bortezomib, CMT1A/1B/2D: Charcot–Marie–Tooth disease type 1A/1B/2D, dnSarm1: dominant negative SARM1, DNA: desoxyribonucleinacid, dSarm: Drosophila sterile alpha and TIR motif–containing protein, EAE: experimental autoimmune encephalomyelitis, HFD: high fat diet, HSP: hereditary spastic paraplegia, IENFD: intraepidermal nerve fiber density, KO: knockout, Nmnat: nicotinamide mononucleotide adenylyltransferase, NMNAT1/2/3: nicotinamide mononucleotide adenylyltransferase isoform 1/2/3, PN: peripheral neuropathy, RNAi: ribonucleic acid interference, Sarm1: sterile alpha and TIR motif–containing protein 1, SCG: superior cervical ganglion, SMA: spinal muscular atrophy, SNAP: sensory nerve action potential, STZ: streptozotocin, Tau: microtubule associated protein tau, Ube4b: ubiquitination factor E4B, VCR: vincristine, WldS: Wallerian degeneration slow protein
References
- Ropper AH, Samuels MA, Klein JP. Adams and Victor’s Principles of Neurology, 10th ed. McGraw Hill Medical; www.AccessMedicine.com.
- Coleman MP, et al. Annu Rev Neurosci. 2010;33:245–267.
- Coleman MP, et al. Nat Rev Neurosci. 2020;21:183–196.
- Conforti L, et al. Nat Rev Neurosci. 2014;15:394–409.
- Hill CS, et al. Neural Regen Res. 2020;15:604–605.
- Gilley J, et al. J Neurosci. 2013;33:9902–9915.
- Milde S, et al. Neurobiol Aging. 2014;35:279–285.
- Dingwall CB, et al. J Clin Invest. 2022;132:e154922.
- Osterloh JM, et al. Science. 2012;337:481–484.
- Gilley J, et al. Elife. 2021;10:e70964.
- Geisler S, et al. Brain. 2016;139:936–952.
- Turkiew E, et al. J Peripher Nerv Syst. 2017;22:90–99.
- Geisler S, et al. JCI Insight. 2019;4:e129920.
- Cheng Y, et al. Diabetes. 2019;68:131–142.
Introduction
- Nerve biopsies have a long tradition beginning at the start of the last century, with procedures performed since the middle of the last century. Although light microscopy may seem outdated, there are still new techniques, and nerve biopsies still play an important role in research. There are possibilities to investigate the fibers by new techniques that may hopefully lead to better understanding.
- Diagnostic approaches to peripheral neuropathy include clinical evaluation, neurophysiology, blood markers, imaging (ultrasound, MRI), and nerve biopsy.
Debate on the need of nerve biopsies: Do we still need nerve biopsies?
- Increasing role of serum markers: Autoantibodies, markers of axonal/myelin damage
- Diagnosis of most autoimmune neuropathies is based on clinical and electrophysiological findings
- Mostly sensory nerves used for biopsies ≥ limited value in motor neuropathies
- Invasive, time-consuming
Nerve biopsies in unclear cases
- In unclear cases, nerve biopsy can help—studies show about 27.8% of patients without any working diagnosis benefited, and in another study 14% saw a diagnostic change1,2.
- Pain is reported post-biopsy, although frequency varies between studies. Nerve biopsy can detect vasculitis, sometimes found unexpectedly in cases not previously suspected2.
Case Study
- A 54-year-old female patient presented with distal symmetric, sensory motor, slowly progressive neuropathy for 8 months. She had similar symptoms before.
- Examination showed absent reflexes, mild toe extensor weakness, symmetric hypesthesia of feet and lower legs.
- CSF was normal; nerve conduction studies showed neuropathy. Nerve biopsy showed signs of loss and degeneration, with inflammatory infiltrates.
- Immunohistochemistry confirmed inflammation. Diagnosis: Vasculitis. She was treated accordingly.
When to perform nerve biopsies
- No fixed rules exist on when to perform nerve biopsy; decisions are individual. One flowchart suggested biopsy when disease is severe, progressive, and asymmetric.3
- Muscle biopsy can be done to increase sensitivity. Ultrasound can guide selection of biopsy site4.
- CSF and nerve MRI can also help differentiate vasculitis from mimics5.
- Findings are not always specific but can help differentiate chronic versus acute pathology6.
Nerve biopsy in the EAN/PNS guidelines of CIDP7
- Suspected CIDP but not confirmed with clinical, laboratory, imaging and electrodiagnostic studies
- Suspected CIDP but no response to treatment (alternative diagnosis?)
- But only when: experienced facilities (surgeons, laboratory, examiners) are available + symptoms severe enough to justify side effects of a biopsy
- Patients need to be informed about the low accuracy of nerve biopsy.
What did we learn from nerve biopsies in the past?8-9
Historically, nerve biopsies provided key knowledge in CIDP and autoimmune neuropathies in the 1980s and 1990s. Biopsy materials helped identify antibodies against paranodal proteins, including contactin-1.8
- Contactin-1 antibody patients showed preserved myelin on MRI but abnormal nodes due to direct attack on motor terminals. Lucky to have biopsy tissue from patients which showed nodal damage in high-resolution microscopy.8
- Other studies from Japan also showed nodal destruction. Research from preserved biopsy tissues provided insights into pathophysiology not visible through routine techniques9.
Future perspectives of peripheral neuropathology10
- Decreasing number of nerve biopsies
- Good storage of biopsy material (biobanking)
- Broad consent for use in research
- Alternative tissue: skin biopsies?
- New techniques: Spatial transcriptomics → tissue needed, Artificial intelligence for analysis and interpretation of tissue sections in the clinical context, Super-resolution microscopy → investigate nerve biopsies on a molecular level
Ultrastructural anatomy of the nodes of Ranvier by STED microscopy11
- Analysis or 12 nodal/paranodal glial and axonal proteins in mouse teased fibers
- Information on the ultrastructure of nodes of Ranvier, organization and periodicity of proteins
dSTORM imaging of nodes of Ranvier of patients with neuropathies12
- 190 nm periodic arrangement of cytoskeletal and axoglial adhesion molecules in human nerves
- Increase of periodicity in patients with peripheral neuropathies (axonal & demyelinating)
- Loss of proteins of the axoglial complex & detachment of the axoglial complex from spectrin-2
dSTORM imaging of nodes of Ranvier of a patient with anti-pan-neurofascin autoantibodies13
- 14% of the nodes severely damaged (conventional fluorescence microscopy)
- Decreased density of NF155, NF186 and Caspr1 even in normal-looking nodes
- Preserved colocalization, normal density of Nav
- Beta-IV-spectrin only altered in severely damaged nodes
Summary: How can nerve/skin biopsies play a role in the future?
- Skin biopsies used for longitudinal assessment but limited in evaluating myelinated fibers. Detection of nodal structures is hard due to few fibers. Requires multiple sections to get enough data. New techniques like spatial transcriptomics, proteomics, super-resolution microscopy are now used in research.
- Skin biopsies may be a future alternative due to ability to repeat and easier access. Essential to store nerve biopsy tissue safely with patient consent in biobanks for future use.
Abbreviations
AINCAT: Adjusted Inflammatory Neuropathy Cause and Treatment score, Caspr1: Contactin-associated protein 1, CIDP: Chronic Inflammatory Demyelinating Polyneuropathy, CSF: Cerebrospinal Fluid, dSTORM: Direct Stochastic Optical Reconstruction Microscopy, EAN: European Academy of Neurology, INC: Inflammatory Neuropathy Consortium, INCAT: Inflammatory Neuropathy Cause and Treatment score, MRI: Magnetic Resonance Imaging, Nav: Voltage-gated Sodium Channel (Nav family), NF155: Neurofascin155, NF186: Neurofascin186, PNS: Peripheral Nervous System, STED: Stimulated Emission Depletion (superresolution microscopy)
References
- Lee H, et al. J Clin Neurosci. 2023;109:1–7.
- Gabriel CM, et al. J Neurol Neurosurg Psychiatry. 2000;69:442–446.
- Nathani N, et al. Muscle Nerve. 2021;64:22–30.
- Costa AF, et al. Ann Rheum Dis. 2024;83:155–163.
- Padilha IG, et al. Neuroimaging Clin N Am. 2024;34:45–60.
- Therapath Neuropathology. Nerve pathology overview. https://therapath.com/nerve-pathology-overview/. Accessed February 2026.
- Van den Bergh PYK, et al. J Peripher Nerv Syst. 2021;26:242–268. / Eur J Neurol. 2021;28:3556–3583.
- Doppler K, et al. Brain. 2016;139:282–295.
- Koike H, et al. J Neurol Neurosurg Psychiatry. 2017;88:1074–1084.
- Ono K, et al. Neuropathol Appl Neurobiol. 2024;50:e12967.
- D’Este E, et al. Proc Natl Acad Sci U S A. 2016;113:132–137.
- Appeltshauser L, et al. Neurobiol Dis. 2023;185:105599.
- Da Cruz Neris R, et al. Front Immunol. 2025;16:152345.
Objective
To explore molecular mechanisms underlying peripheral nerve repair, particularly focusing on:
- Schwann cell responses to nerve injury
- The role of Neuregulin-1 (Nrg1) signaling
- Mitochondrial metabolic adaptations
- Leptin receptor signaling in regeneration
- Translational implications for hereditary and acquired neuropathies (e.g., CMT1A, CMT1B, obesity-related neuropathy)
Schwann Cell Repair Program in Acute Injury1-2
After acute nerve injury, Schwann cells undergo adaptive reprogramming into a "repair Schwann cell" state, enabling nerve regeneration.1
- These repair Schwann cells break down myelin breakdown (via autophagy) and facilitate axonal regrowth via bands of Büngner.1
- Unlike development, where axons drive Schwann cell maturation, injury triggers glial-derived neuregulin-1 (NRG1) signaling, essential for repair. 2
Dysregulated Repair in Neuropathies3-4
In CMT1A (demyelinating neuropathy), Schwann cells exhibit a chronic, maladaptive repair response, leading to onion bulb formations (pathological hallmark).3
- NRG1 signaling, beneficial in acute injury, becomes pathogenic in CMT1A, driving supernumerary Schwann cell survival and concentric remodeling.3
- CMT1B (late-onset demyelination) shows delayed repair activation; NRG1 loss worsens pathology, suggesting context-dependent roles.4
- Nrg1 signaling correlates with disease progression in CMT1A and CMT1B mouse models and is elevated in human biopsies.4
Metabolic Demands of Repair3-4
- Schwann cells increase mitochondrial activity and oxidative phosphorylation during repair, fueled by fatty acids from myelin breakdown.3
- Leptin receptor signaling (induced post-injury) regulates mitochondrial expansion and energy metabolism.4
- Leptin receptor knockout impairs axon regeneration and remyelination after injury.4
Therapeutic Implications3
- Leptin treatment post-injury improved electrophysiological and histological outcomes in mice.
- In obesity-related neuropathy, hyperleptinemia exacerbates pathology; leptin receptor ablation in Schwann cells reduced neuropathic symptoms.
Conclusion
- The Schwann cell repair program is optimized for transient injury but becomes harmful when chronically activated (e.g., in CMT).
- Dual role—promotes regeneration in injury but drives pathology in demyelinating neuropathies.
- Targeting intrinsic (Nrg1) and extrinsic (leptin receptor) pathways offers therapeutic potential, but strategies must differentiate between acute injury and chronic disease states.
- Leptin links adipose tissue to Schwann cell metabolism, offering a therapeutic target for neuropathies (e.g., obesity-related or inherited).
- Interventions must balance repair activation (to aid remyelination) versus suppression (to prevent chronic damage).
Abbreviations
CMT: Charcot Marie Tooth disease, CMT1A: Charcot Marie Tooth disease type 1A, CMT1B: Charcot Marie Tooth disease type 1B, Nrg1 / NRG1: Neuregulin 1
References
- Stassart RM, et al. Glia. 2018;66:151–167.
- Stassart RM, Fledrich R, et al. Nat Neurosci. 2013;16:889–897.
- Fledrich R, Stassart RM, et al. Nat Med. 2014;20:1055–1061.
- Fledrich R, Akkermann D, et al. Nat Commun. 2019;10:1–15.
Objective
- To highlight the need for continued progress in CIDP management, emphasizing that despite over 25 years of recognition, not much has been done and more needs to be done, with a clear distinction between the goals of induction treatment and remission maintenance.
Treatment approaches
- CIDP treatment has evolved from steroid-responsive therapy in 1958 to IVIG and plasma exchange in the 1980s, SCIG in 2018, and is now progressing toward targeted inhibitors of FcRn, complement, and BTK by 2024.1
- Distinguished between induction treatment (aimed at achieving initial improvement) and maintenance treatment (focused on sustaining remission).2
Clinical Trial Insights
- Analysis of IVIg vs placebo showed 55% improvement with IVIG vs 23% with placebo. Risk Ratio: 2.40 (CI: 1.72–3.35). Comparison of IVIG vs prednisolone showed no significant difference (Risk Ratio: 0.91)3.
- In the ICE trial (INCAT-DS), 30 IVIG responders were identified, with most showing improvement within 6 weeks4,5.
- Time to Remission (Dexamethasone vs Prednisolone): Dexamethasone: Median 20 weeks to remission. Prednisolone: Median 39 weeks. Improvement with steroids is slower compared to IVIG in the ICE trial6.
- Steroid Remission Outcomes (PREDICT Study): trial with 40 patients comparing dexamethasone and prednisolone. Remission at 1 year: ~40%. Remission at 4.5 years: 26%6,7.
- The OPTIC trial evaluated IVIG plus intravenous methylprednisolone (IVMP) vs. IVIG alone in untreated CIDP patients, finding no significant difference in 1-year remission rates but showing greater short-term functional improvements with combination therapy; however, safety concerns (thrombotic events) led to early termination8.
- Trial focused on induction treatment strategies in CIDP: Combination of IVIG and steroids may offer faster improvement (IVIG) and potentially longer remissions (steroids) but poses safety concerns like thrombosis; the ICE 2008 trial showed IVIG reduces relapse by 40%, yet 50% of placebo patients remained relapse-free for 6 months, raising questions on treatment duration and patient selection4.
- In CIDP maintenance, uncertainties persist around overtreatment (seen in 20–40% of cases), optimal timing to taper or stop treatment, and how reliably and rapidly patients can restabilize—though most restabilize fully and quickly. In the IVIG withdrawal, 60 stable patients were randomized; placebo led to 60% relapse versus 40% in the IVIG group, with only 28% in remission after one year, highlighting the challenge of discontinuing therapy and the need for biomarkers to guide maintenance9.
Abbreviations
BTK: Bruton's Tyrosine Kinase, CI: Confidence Interval, CIDP: Chronic Inflammatory Demyelinating Polyneuropathy, FcRn: Neonatal Fc Receptor, ICE: Intravenous Immune Globulin for the Treatment of CIDP Efficacy Trial, INCATDS: Inflammatory Neuropathy Cause and Treatment – Disability Score, IV: Intravenous, IVIG: Intravenous Immunoglobulin, IVMP: Intravenous Methylprednisolone, SCIG: Subcutaneous Immunoglobulin
References
- Mair MJ, et al. J Neurol Neurosurg Psychiatry. 2025;96.
- Van den Bergh PYK, et al. J Peripher Nerv Syst. 2021;26:242–268 / Eur J Neurol. 2021;28:3556–3583.
- Eftimov F, et al. Cochrane Database Syst Rev. 2013;2013:CD006459.
- Hughes RAC, et al. Lancet Neurol. 2008;7:136–144.
- Latov N, et al. Arch Neurol. 2010;67:1036–1041.
- van Schaik IN, et al. Lancet Neurol. 2012;11:493–502.
- Eftimov F, et al. Neurology. 2012;79:188–193.
- Nobile-Orazio E, et al. Lancet Neurol. 2012;11:493–502.
- Adrichem ME, et al. Brain. 2022;145:2345–2356.
Agenda
- To summarize recently completed and ongoing trials in CIDP
- To recognize challenges in CIDP trial design
- To recognize challenges for future CIDP trial logistics
The pre-ICE1 age: Lessons learned
- Randomized placebo-controlled trials are needed
- To perform an adequately power prospective RCT, multicenter multinational collaboration is needed
- With recognition of CIDP as a syndrome, a thoughtful approach to inclusion criteria is needed
The post-ICE1age
| The ICE Trial (Year: 2008)1 | The PATH Trial (Year: 2018)2 | The FORCIDP Trial (Year: 2018)3 | The ProCID Trial (Year: 2022)4 |
|---|---|---|---|
| General Design | General Design | General Design | General Design |
| Status: Naïve or off treatment | Status: On IVIG | Status: On IVIG or corticosteroids | Status: On IVIG or corti costeroids (washed out) |
| Randomized: IVIG or Placebo | Randomized: SCIG or Placebo | Randomized: Fingolimod or Placebo | Randomized: IVIG (Multiple dose) |
| Outcome: Response rate 24 weeks | Outcome: Relapse rate 24 weeks | Outcome: Relapse rate 24 weeks | Outcome: Response rate 24 weeks |
| The ADVANCE Trial (Year: 2023)5 | My CIDP Choice trial (Year: 2024)6 | The ADHERE Trial (Year: 2024)7 | CIDPRIT Trial (Year: 2025)8 |
|---|---|---|---|
| General Design | General Design | General Design | General Design |
| Status: On IVIG | Status: On IG | Status: Most on IVIG or corticosteroids | Status: On IVIG |
| Randomized: fSCIG or Placebo | Randomized: Rozanolixizumab or Placebo | Randomized: Efgartigimod or Placebo | Randomized: Rituximab or Placebo |
| Outcome: Relapse rate 32 weeks | Outcome: Relapse rate 13 weeks | Outcome: Relapse rate 48 weeks | Outcome: Relapse at 12 weeks |
Placebo response rates9: Measuring improvement vs relapse.
Two clinical trial scenarios:
- First: Off treatment randomized to study drug or placebo measuring improvement
- Second: On treatment randomized to study drug or placebo measuring relapse
Clinical trials that measure improvement seem less prone to placebo effect than those that measure relapse or worsening.
Summary of post-ICE age trials2-7
| PATH2 2018 | FORCIDP3 2018 | ProCID4 2022 | ADVANCE5 2023 | MyCIDP6 2024 | ADHERE7 2024 | |
|---|---|---|---|---|---|---|
| Adjudication panel | No | No | No | No | No | Yes |
| Dependency phase | Yes | No | Yes | No | No | Yes |
| Primary improvement or relapse | Relapse | Relapse | Improve | Relapse | Relapse | Relapse |
| Included Treatment naïve | No | No | No | No | No | Yes |
Dependency phase and Adjudication panel
Three studies with trial data show improvements in patients, though some improvements are small and not widely included.
- The question is where the adjudication panel fits within the dependency phase. Looking at placebo effects in various trials, around 37 to 46% of patients in randomized trials did not get worse, even with the dependency phase attempting to sort out patients.4
- In comparison, trials without adjudication panels had even higher percentages of patients not worsening, between 60 and 70%.4
- The dependency phase with adjudication panels removed only about 11% of patients during withdrawal because they did not get worse.4
- In one study, only 2% failed the dependency phase and relapsed. This raises questions about the value of the dependency phase, considering the effort, cost, and anxiety it causes clinicians and patients.7
- Despite these challenges, some trials that included advanced adjudication were successful.7
Lessons learned so far
- Not very good at assessing disease activity4
- Although dependency phases and adjudication panels are not perfect, recent trials without dependency confirmation have had very high placebo response results2,4
- With a parallel group withdrawal design, the margin to detect benefit is narrow7
Trial designs of the following trials were presented:
INVGOR Study – Open label, sequential dose cohort10
- Drug: NVG-2099, a recombinant Fc fragment immunomodulator
- Cohorts: Three cohorts with different dosing and patient experience levels
- Duration: Screening (6 weeks), Treatment (14 weeks), Follow-up (6 weeks)
CASCA Study – Open label, single group assignment11
- Drug: TAK-411, Hypersialylated Immunoglobulin G
- Dosing: TAK-411 400 mg/kg IV, with potential repeat after 3 weeks
- Outcome: Stability in INCAT at week 24
TAK-881 Study – Open label12
- Drug: TAK-881, next-generation IgG + hyaluronidase
- Dosing and Phases: Includes ramp-up and treatment epochs with fSCIG and TAK-881
- Outcome: Primary focus on total IgG levels; secondary on relapse rate and grip strength
IMVT-1401-2401 Trial – Multistage, double-blinded, placebo-controlled withdrawal13
- Drug: Batoclimab, an FcRn antagonist
- Outcome: Relapse rate in Period 2
CAPTIVATE Trial – Randomized withdrawal, double-blind, placebo-controlled14
- Drug: DNTH-103, an aC1s inhibitor
- Outcome: Response improvement in Part A, efficacy (time to relapse) in Part B
ARISE Trial – Withdrawal15
- Drug: Nipocalimab, an FcRn antagonist
- Phases: Includes stages A and B with open-label and randomized components
- Outcome: Percent responders and time to relapse
VITALIZE Study – Randomized, double-blinded, controlled16, 17
- Drug: Riliprubart, a C1s inhibitorRiliprubart, a C1s inhibitor
- Outcome: Percent of patients improving (aINCAT) at week 24
EMVIGORATE Study – Randomized, double-blinded, controlled18
- Drug: Empasiprubart, a C2 inhibitor
- Outcome: Percent of patients improving (aINCAT) at week 24
Final thoughts & Summary
- Accurate diagnosis and measuring treatment affect are ongoing challenges for clinical trials
- Optimization of adjudication panels and dependency analysis may help
- Designs that shift toward measuring response rather than relapse may help
- Biomarkers or clinical tools to more reliably detect disease activity (or no evidence of disease activity) are needed
- Finding patients and investigators to sustain trial growth will be a challenge over upcoming years
Abbreviations
aINCAT: Adjusted Inflammatory Neuropathy Cause and Treatment score, CIDP: Chronic Inflammatory Demyelinating Polyneuropathy, C1s: Complement component 1s, C2: Complement component 2, Fc: Fragment crystallizable region, FcRn: Neonatal Fc receptor, fSCIG: Facilitated Subcutaneous Immunoglobulin, GBS: Guillain-Barré Syndrome, ICE: Intravenous Immune Globulin in CIDP Efficacy trial, IG: Immunoglobulin, INCAT: Inflammatory Neuropathy Cause and Treatment score, IV: Intravenous, IVIG: Intravenous Immunoglobulin, RCT: Randomized Controlled Trial, SCIG: Subcutaneous Immunoglobulin
References
- Hughes RAC, et al. Lancet Neurol. 2008;7:136–144.
- Van Schaik IN, et al. Lancet Neurol. 2018;17:405–415.
- Hughes RAC, et al. Lancet Neurol. 2018;17:374–383.
- Cornblath DR, et al. Brain. 2022;145:2169–2180.
- Bril V, et al. J Peripher Nerv Syst. 2023;28:123–134.
- Querol L, et al. J Neurol Neurosurg Psychiatry. 2024;95:450–458.
- Allen JA, et al. Lancet Neurol. 2024;23:512–523.
- Nobile-Orazio E. Brain. 2024;147:284–296.
- Lewis RA, et al. J Peripher Nerv Syst. 2020;25:63–72.
- ClinicalTrials.gov. NCT07027111. https://clinicaltrials.gov/NCT07027111 (accessed May 2025).
- ClinicalTrials.gov. NCT06798012. https://clinicaltrials.gov/NCT06798012 (accessed May 2025).
- ClinicalTrials.gov. NCT06747351. https://clinicaltrials.gov/NCT06747351 (accessed May 2025).
- ClinicalTrials.gov. NCT05581199. https://clinicaltrials.gov/NCT05581199 (accessed May 2025).
- ClinicalTrials.gov. NCT06858579. https://clinicaltrials.gov/NCT06858579 (accessed May 2025).
- ClinicalTrials.gov. NCT05327114. https://clinicaltrials.gov/NCT05327114 (accessed May 2025).
- ClinicalTrials.gov. NCT06290141. https://clinicaltrials.gov/NCT06290141 (accessed May 2025).
- ClinicalTrials.gov. NCT06290128. https://clinicaltrials.gov/NCT06290128 (accessed May 2025).
- ClinicalTrials.gov. NCT06920004. https://clinicaltrials.gov/NCT06920004 (accessed May 2025).
Background
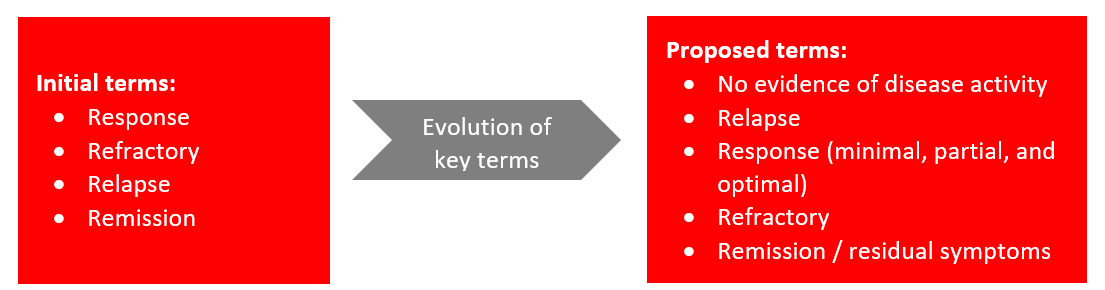
- The terms Response, Refractory, Relapse, and Remission are utilized differently by different stakeholders in CIDP—patients, clinicians, payers, and regulators—leading to confusion, miscommunication, and potential barriers to treatment access and evaluation of therapies.
- Over the past year, a leadership collaborative supported by the GBS/CIDP Foundation International has been working to build a common definition related to CIDP and conducting workshops1.
- Key takeaways from patients’ workshops (n=16)
- General feedback: Workshop participants were skeptical about the key clinical terms presented. Definitions should be reworked to better reflect patients' lived experiences.
- Definition feedback
- Response: Patients emphasized that clinicians need to be more attuned to both positive and negative changes they experience. They expressed frustration at having to convince their doctor that a change has occurred. Participants identified successful treatment as the ability to live independently.
- Refractory: Patients don't want the word "refractory" to provide an excuse to give up.
- Remission: The remission definition was problematic for CIDP participants. Some felt it meant a lack of flares or presence of disease while continuing treatment, while others described it as occurring after treatment was discontinued. Participants also noted that some people will have residuals for the rest of their lives.
- Relapse: Distinguish relapse from flare. Clarify time or severity components.
- Key takeaways from patients’ workshops (n=16)
- The goal was to standardize definition to improve communication and care.
Consensus workshop objectives and process overview1
- The project team, representing the leadership collaborative, met in April to review all feedback from patient workshop.
Objective
- Reach consensus on how to employ definitions across patient care and clinical research
- Develop a strategy for broad acceptance of these definitions across all key stakeholders
- Process overview
- Used a modified approach to reach consensus. The steering committee reviewed relevant literature, drafted a definition, and revised it after discussion.
- The larger group then reviewed and voted on the definition. Since consensus wasn’t reached initially, panel members discussed key points, followed by group discussions and repeated voting until consensus was achieved.
Consensus definitions & considerations
- Participants voted on a 1–9 scale: 1–3 meant strong disagreement, 4–6 meant partial agreement, and 7–9 meant agreement with most of the definition. A score of 7 or higher was required for agreement, and consensus needed 80% of voting members to score 7 or above.
Consensus level of agreements
Immune Neuropathy - No Evidence of Disease Activity (IN-NEDA): Consensus (7 or above): 95% agreement (n=20); Revisions: 1
- Definition components: 1) Patient Global Impression of Change (PGIC); 2): Disability metric defined as a I-RODS2 4-point (centile) change or INCAT3 1-point change; 3) Impairment metric: Defined as a change in grip strength (8 kPa or 10% averaged change) and MRC 60 sum score (2 points).
- Proposed IN-NEDA classifications:
- IN-EDA: Patient perception of change AND a change in disability, impairment, or both. There must be no other condition that better explains change in disability and impairment.
- IN-NEDA 1: No patient perception of change AND either no decline in Disability OR no decline in Impairment for a duration of 6 months or more.
- IN-NEDA 2: No patient perception of change AND no decline in Disability AND no decline in Impairment components for a duration of 6 months or more.
- Recommendation for clinical trials: target NEDA-2 status
Relapse: Consensus: 100% agreement (n=20); Revisions: 1
- Relapse: Patient perception of change AND decline in disability AND impairment fulfilling IN-EDA definition following a period of stability or improvement, in the absence of a condition that better explains change in function and impairment, over a period of 12 weeks or less.
- Possible Relapse: Patient perception of change AND decline in disability OR impairment fulfilling IN-NEDA1 definition following a period of stability or improvement, in the absence of a condition that better explains change in function and impairment, over a period of 12 weeks or less.
Response: Minimal / Partial / Optimal
- Minimal response: Patient perception of improvement AND a minimum change on at least one outcome measure suggesting at least early partial response to a specific treatment, assessed within 12 weeks.
- Improvement = CIDP Guideline recommendations, minimum range
- INCAT 1, I-RODS centile 4, MRC 2, GS 8 kPa
- Consensus: 90% agreement (n=20); Revisions: 1
- Partial response: Patient perception of improvement AND a change on at least one outcome measure within 24 weeks.
- Improvement = CIDP Guideline recommendations, maximum range
- INCAT 1, I-RODS centile 8 (or reaching maximum), MRC 4, GS 14 kP
- Consensus: 100% agreement (n=19); Revisions: 1
- Optimal response: Recovery as close as possible to the pre-CIDP functional status excluding potential residuals and fulfilling IN-NEDA 2 criteria sustained for at least 6 months.
- Consensus: 100% agreement (n=19); Revisions: 2
- Minimal response: Patient perception of improvement AND a minimum change on at least one outcome measure suggesting at least early partial response to a specific treatment, assessed within 12 weeks.
Refractory: Consensus: 90% agreement (n=20); Revisions: 1
- Absence of minimal response OR relapse leading to/requiring treatment discontinuation after adequate dosing and duration of the administered therapy.
Remission: Consensus: 100% agreement (n=19); Revisions: 1
- No signs of disease activity (IN-NEDA 2) with or without residuals without immunosuppressive/immunomodulatory therapy for at least 12 months.
- Residuals: Consensus: 95% agreement (n=19); Revisions: 1
- Signs, symptoms, or impairments related to CIDP that persist despite adequate immunosuppressive/immunomodulatory therapies and are present in a patient who otherwise has no evidence of disease activity (IN-NEDA).
Conclusion
- Patients, clinicians, researchers, regulatory agencies, industry, and payers need standardized key terms for use in clinical practice and clinical trials.
- The consensus-driven definitional blueprint aims to improve management of CIDP, quality of clinical trials, and patient experience.
- The results, along with additional details on the group's deliberations, will be addressed in a forthcoming paper.
- More work is needed to guide adoption of these consensus-derived terms in practice & research
- These terms will also be incorporated into a visual patient journey map with lay language to improve comprehension and awareness of the definitions
- Unmet needs
- The lack of biomarkers is a critical gap in defining key clinical terms in CIDP
- The definitions will continue to evolve as new biomarkers and treatments become available
Abbreviations
CIDP: Chronic Inflammatory Demyelinating Polyneuropathy, GBS: Guillain-Barré Syndrome, GS: Grip Strength, IRODS: Inflammatory Rasch-Built Overall Disability Scale, INCAT: Inflammatory Neuropathy Cause and Treatment Disability Score, INEDA: Immune Neuropathy – Evidence of Disease Activity, INNEDA 1/2: Immune Neuropathy – No Evidence of Disease Activity Category ½, kPa: Kilopascal, MRC: Medical Research Council Sum Score, PGIC: Patient Global Impression of Change
References
- GBS|CIDP Foundation International. Leadership Collaborative. https://www.gbs-cidp.org/gbscidp-leadership-collaborative/ (accessed Sept 2025).
- van Nes SI, et al. Neurology. 2011;76:337–345.
- Breiner A, et al. Muscle Nerve. 2014;49:246–252.
Symposia Highlights
Agenda & Objectives
- Reflect on the day-to-day challenges patients face, and the unmet needs with current standard treatment approaches
- Explore emerging innovative and targeted treatment approaches with potential to improve patient outcomes
1) Exploring disease burden and unmet treatment needs in CIDP
- Patients with CIDP face significant burden: impaired quality of life (~60% issues with activity1,2, 27% anxiety/depression2), disability (~10% wheelchair3, ~60% mobility problems1,2), neuropathic pain (20%), residual pain (52%), and fatigue (77%) post-treatment4,5.
- Diagnosis delays median 9 months, with ~20% underdiagnosis6, inconsistent diagnostic criteria use7, neurophysiology misinterpretation6, and 32–47% overdiagnosis6,8.
- Unmet needs include treatment inconvenience9,10, side effects10-12, limited response (~30% complete response)13, residual disability14, persistent symptoms10, and absenteeism15.
- Monitoring challenges involve assessing disease activity16, treatment decisions16, and relapse prediction16,17.
Rationale of existing immunotherapies:
- IVIG has a 70% response rate with side effects like headache, fever, hypertension, and rare thromboembolism; SCIG increases patient autonomy18.
- Corticosteroids are effective but take longer to improve and relapse, with more side effects than IVIG18.
- PLEX offers short-term improvement but risks rapid deterioration, requires central access, hospital stays, and carries risk of fatal adverse events18.
- Existing immunotherapies lack strong efficacy evidence due to significant trial design limitations18.
2) Evaluating emerging targeted therapies for CIDP
- The EAN/PNS guidelines provide a structured approach to CIDP treatment, starting with IVIG and corticosteroids, and considering diagnosis reassessment before escalating to off-label immunotherapies19.
- B-cell depleting therapy with rituximab showed ~60% benefit in case series20, but clinical trials (Japanese21 and Italian22) failed to meet primary endpoints. Complement inhibitors are under investigation: CAPTIVATE trial (C1s), an ongoing C2 inhibitor trial, and a Phase 2 open-label study showing high stability/improvement rates across standard, refractory, and naive cohorts23. Phase 3 trials (MOBILIZE24/VITALIZE25) are testing similar endpoints.
- FcRn antagonists (Efgartigimod26, Nipocalimab28, Rozanolixizumab27) block IgG recycling; ADHERE showed 66-70% response and a 61% relapse risk reduction26, while the Rozanolixizumab trial was negative27. Key issues remain in patient selection and transition strategies26,28.
Patient profile: Difficult to treat
- A 76-year-old man with rapid proximal and distal weakness, absent leg reflexes, normal sensory function, elevated CSF protein, and abnormal motor studies was diagnosed with CIDP.
- Initial IVIG treatment improved strength temporarily, but deterioration followed.
- Repeat neurophysiology confirmed typical CIDP features. Plasma exchange and steroids led to significant improvement and near-normal function.
- The case highlights diagnostic challenges, treatment adjustments, limited biomarkers, and the use of patient outcomes and objective measures to guide therapy.
Summary
- CIDP patients face significant disease burden and disability, with delays and inaccuracies in diagnosis impacting care. Current immunotherapies have limitations including side effects and incomplete efficacy.
- There is an unmet need for more effective, targeted, and safer treatments. Improved monitoring tools and standardized diagnostic criteria are essential to optimize treatment and manage progression.
- Emerging targeted therapies and patient-centric clinical trials offer promise. Diagnosis is challenging without definitive biomarkers, requiring individualized treatment and clinical judgment to avoid over- or under-treatment.
Abbreviations
CIDP: Chronic Inflammatory Demyelinating Polyneuropathy, CSF: Cerebrospinal Fluid, EAN: European Academy of Neurology, FcRn: Neonatal Fc Receptor, IgG: Immunoglobulin G, IVIG: Intravenous Immunoglobulin, PNS: Peripheral Nerve Society, PLEX: Plasma Exchange, SCIG: Subcutaneous Immunoglobulin
References
- Mahdi-Rogers M, et al. Eur J Neurol. 2014;21:34–39
- Bus SRM, et al. J Neurol. 2022;269:945–955
- Hafsteinsdottir B Olafsson E. Eur J Neurol. 2016;75:263–268
- Bjelika B, et al. J Peripher Nerv Syst. 2019;24:180–186
- Busnotchen C, et al. J Peripher Nerv Syst. 2019;24:253–259
- Broers M, et al. Eur J Neurol. 2021;28:2065–2072
- Gelinas D, et al. J Neurol Sci. 2019;397:84–91
- Allen JA, Neurol Ther. 2020;9:43–54
- Watkin JM, et al. J Neurosci Nurs. 2019;51(4):198-203
- Querol L, et al. J Neurol. 2020;267:3706–3716
- Dalakas M. Pharmacol Ther. 2004;102:177–193
- Choi K, et al. Front Neurol. 2024;15:1456835
- Rajabally YA, et al. Eur J Neurol. 2024;31:e16399
- Allen JA, et al. Lancet Neurol. 2024;23:1013–1024
- Allen JA, et al. Adv Ther. 2021;38:316–328
- Fisse AL, et al. Neurol Res Pract. 2020;2:42
- Bus SRM, et al. J Neurol. 2022;269:945–955
- Mair D, et al. J Neurol Neurosurg Psychiatry. 2025;96:38-46
- Van den Bergh PYK, et al. J Peripher Nerv Syst. 2021;26:242–268
- Chaganti S, et al. Neuromuscul Disord. 2022;32:621–7
- Shimizu S, et al. JMIR Res Protoc. 2020;9(4):e17117
- Nobile-Orazio E, et al. Brain. 2025;148:11 12–21
- Dianthus Therapeutics. Corporate presentation. March 2025
- https://clinicaltrials.gov/study/NCT06290128 (accessed May 2025)
- https://clinicaltrials.gov/study/NCT06290141 (accessed May 2025)
- Allen JA, et al. Lancet Neurol. 2024;23:1013–24
- Querol L, et al. J Neurol Neurosurg Psychiatry. 2024;95:845–54
- Levine T, Muley S. J Neurol Sci 2024 468(10):123313
Agenda & Objectives
- To understand the evidence supporting CIDP management to enhance clinical knowledge and practice
- To explore strategies for integrating evidence-based practices into clinical decision-making to help improve patient outcomes
- To learn from case studies and expert insights to navigate common challenges in CIDP management, with the goal of delivering improved patient care and achieving better clinical outcomes
Integrating evidence and experience into CIDP care
Case study 1: A 56-year-old from a remote area in British Columbia experiences progressive motor and sensory symptoms.
- Diagnostic tests showed demyelination and conduction block, indicating typical CIDP.
- A survey was conducted and most of the audience selects IVIG as the initial treatment, but the patient was treated with dexamethasone due to geographic and logistic constraints.
- He showed significant improvement and returns to work
Therapies recommended by the EAN/PNS 2021 guidelines for the treatment of CIDP:Three first-line CIDP treatments: plasma exchange, corticosteroids, and IVIG were discussed
- Plasma Exchange1: Effective but less commonly used due to access and side effects.
- Five exchanges over 2 weeks; Central line often required for induction therapy; Maintenance therapy tailored to patient, peripheral lines if possible
- Corticosteroids1: Several regimens exist. Real-world evidence shows efficacy, particularly with pulse regimes, though not recommended for motor-predominant CIDP.
- Daily prednisone or prednisolone 1-1.5 mg/kg, at least 6 weeks, then slow taper over ≥8 months
- Monthly oral pulsed dexamethasone 40 mg daily, 4 days per month for 6 months
- Monthly IV pulsed methylprednisolone 500 mg daily, 4 days, then 1-2g/month thereafter for 6 months
- IVIG2: Strong evidence from randomized trials, with rare serious side effects.
- Panelists from the survey weigh factors such as cost, logistics, remission potential, and patient preferences when choosing a treatment.
- Clinicians stress individualized decisions based on efficacy, access, patient preference, and side effect profiles.
Case study 2: A 60-year-old healthcare worker with mild to moderate CIDP and a history of osteopenia prefers avoiding corticosteroids. She was started on IVIG, improved significantly by 4 months, and returned to work.
- Ongoing Treatment Discussion: A range of treatment continuation strategies were debated
- Options include stopping IVIG, tapering, continuing for a fixed duration (e.g., one year), or ongoing treatment to prevent relapse.
- Audience responses from the survey were mixed, reflecting diverse clinical practices.
IVIG for longer-term treatment of CIDP:
ICE Study 2008, maintained response to IVIG over 6 and 12 months3
When to stop first-line treatment: 80% response rate to IVIG at 18 weeks (based on knowledge and clinical experience of speaker). Good practice point: if the first-line treatment is effective, continuation should be considered until the maximum benefit has been achieved1.
Poll panel preferences: One panelist prefers immediate cessation of IVIG with the option to restart quickly if needed. Others favor a fast taper or interval extension to reduce relapse anxiety. Emphasis is placed on managing patient expectations early, clarifying that CIDP treatment is not necessarily indefinite.
Case study 3: A 40-year-old male started with pulse methylprednisolone but switched to IVIG due to psychological side effects and lack of improvement. Responded well to IVIG (100g every 5 weeks) but disliked the impact on work and migraines. Switched to facilitated SCIG (fSCIG), self-administered every 4 weeks (80g/840ml), which he preferred. Patient reported greater flexibility with fSCIG and fewer peak-related side effects, though it caused abdominal swelling and mild skin reactions.
fSCIG vs conventional SCIG:
- Conventional SCIG: Lower bioavailability (~2/3 absorption), weekly smaller doses4.
- fSCIG: Uses hyaluronidase for faster, larger volume infusions, higher bioavailability4.
The ADVANCE-CIDP 1 trial5: A Phase 3, double-blind, placebo-controlled trial conducted at 54 sites in 21 countries. The trial evaluated fSCIG efficacy and safety in preventing CIDP relapse.
- fSCIG reduced relapse (9.7% vs. 31.4% placebo); some placebo patients may have been in remission.
- fSCIG was well-tolerated with mild local reactions and fewer headaches than IVIG.
- Most patients preferred fSCIG over IVIG.
The ADVANCE-CIDP 3 trial6: An open-label extension (33 months median): fSCIG safe and effective (4.5% annual relapse rate); 91% of eligible patients chose to continue the study.
Summary
- Clinical use of fSCIG: Best suited for high-dose patients wanting more autonomy/lifestyle flexibility. Training like SCIG with 3–4 nurse visits plus ongoing support.
- Switching is patient-specific: Some prefer hospital IVIG, others value independence.
- Monitoring Tools: Patients are followed clinically using PROs (R-ODS, fatigue, QoL, pain scores), objective tools (grip strength, MRC sum score, 10m walk, 9-hole peg test). EMG/NCS is used selectively for relapse assessment, pre-tapering, or diagnosis (not routinely).
- Relapse Management: If relapse occurs after remission, restart previous treatment, possibly without full induction if relapse is mild. CIDP relapses vary where some are progressive, others relapsing-remitting.
- Biomarkers: No reliable biomarkers to predict remission. Higher filament levels may indicate poorer outcomes but not routinely used.
- CIDP Variants & Treatment Decisions: Clinical features and variants influence treatment. For multifocal CIDP, evidence is conflicting. IVIG dependency more common. For pure motor CIDP, IVIG preferred. Steroids often tried first in less disabling multifocal cases.
Abbreviations
CIDP: Chronic Inflammatory Demyelinating Polyneuropathy, EAN: European Academy of Neurology, EMG: Electromyography, fSCIG: Facilitated Subcutaneous Immunoglobulin, IV: Intravenous, IVIG: Intravenous Immunoglobulin, MRC: Medical Research Council, NCS: Nerve Conduction Study, PNS: Peripheral Nerve Society, PROs: Patient-Reported Outcomes, QoL: Quality of Life, R-ODS: Rasch-built Overall Disability Scale, SCIG: Subcutaneous Immunoglobulin
References
- Van den Bergh PYK, et al., J Peripher Nerv Syst. 2021;26(3):242-268
- Bus SRM, et al, Cochrane Database Syst Rev, 2024;2(2):CD001797
- Hughes RAC, et al., Lancet Neurol, 2008;7:136-144
- Hadden RDM, et al., touchREVIEWS in Neurology. 2025;21(1):63-72
- Bril V, et al, J Peripher Nerv Syst, 2023;28(3):436-449
- Hadden RDM, et al, J Peripher Nerv Syst, 2024;29(4):441-452
Agenda & Objectives
- The educational symposium provided a focused review of mechanisms of action of key CIDP therapies, highlighting the disease’s heterogeneity and need for better patient profiling to enable personalized, mechanism-based treatment and move beyond trial-and-error approaches.
1) Mechanisms of action of key therapies for CIDP
- CIDP is a heterogeneous disorder comprising diverse underlying mechanisms (autoantibodies, complement activation, T-cell Infiltration, B-cell, etc.) and clinical variants1.
- While segmental demyelination is the pathological hallmark of CIDP, diagnosis relies primarily on electrophysiological testing, as biopsies are rarely performed1.
- Absence of validated biomarkers hampers the ability to align patients with optimal therapies, leading to trial-&-error treatment approaches that may not reflect underlying disease drivers1.
- Therapeutic landscape summary presented at the meeting:
| Therapy | Mechanism | Response |
|---|---|---|
| IVIG | Pleiotropic: autoantibody neutralization, Fc receptor modulation, inhibits complement | ~37-70% patient response (depending on dose, frequency and treatment scheme)2-6 |
| Plasma exchange | Removes pathogenic antibodies (IgG, IgM), complement factors, immune complexes, inflammation mediators | ~50% response; especially useful in removing humoral factors in blood (IgGs, IgM, cytokines complement); associated logistical implications7,8 |
| Corticosteroids | Anti-inflammatory (lyses immune cells, reduces antibodies) | ~60% patient response1,9 |
| FcRn blocker | Reduces IgG levels | ~Two-third of patients improve with efgartigimod in a selected CIDP population1,10 |
| Complement inhibitors | Blocks complement-mediated nerve damage (prevent MAC pore appearance), reduces inflammation | In open label trials, response varied between ~50–88%, depending on CIDP sub-type1,11 |
| Immunosuppressant drugs | Reduced activation of immune cells, apoptosis of inflammatory cells | Variable response rates. Case series and anecdotal cases1,12-14 |
| B-cell Depletion | Eliminates B cells, limits antigen presentation, reduce autoantibodies | Multiple open-label case series showing anecdotal benefit15-17 |
Summary
- The current trial-and-error approach to treating CIDP underscores a critical need for precision medicine.
- Profiling patients on their predominant pathophysiology can enable more effective, individualized treatment.
- Leveraging emerging therapies and technologies to better phenotype patients (clinically and pathophysiologically) presents an opportunity to shift from one-size-fits-all strategies toward more personalized care.
2) Tailoring CIDP therapies: Optimizing care with patients in mind
Objective
- The symposium provided a practical clinical perspective on how to optimize CIDP treatment by minimizing trial-and-error approaches, individualizing therapy based on patient characteristics, and incorporating simple monitoring tools and emerging biomarkers.
Key highlights
- Getting it right first time:
- Accurate diagnosis is essential. Clinical history and examination remain the foundation of patient assessment. In a retrospective review study, Twenty-seven patients (47%) failed to meet minimal CIDP diagnostic standards18.
- Misdiagnosis of CIDP often results from misinterpretation of neurophysiology, particularly the use of the term “demyelination” instead of “conduction slowing,” which may also be seen in conditions like diabetes18.
- The use of red flags, such as TTR mutations or distal phenotypes, can help raise suspicion for alternative diagnosis20.
- Sensory nerve biopsy is suggested in cases when two first-line treatments have failed, when tissue diagnosis is needed to confirm an alternative condition, or when it's unclear whether the patient has anti-MAG neuropathy or CIDP20.
- Treatment strategies
- IVIG (89% respond after 3 doses)21, corticosteroids (Long term remission achieved following 1 or 2 courses of pulsed dexamethasone or an 8-month regimen of daily prednisolone22, and plasma exchange (various regimens) remain first-line therapies (personal choice).
- Sudden cessation vs. gradual tapering shows similar outcomes, but sudden cessation identifies non-responders faster (~17 vs. 40 weeks)23. ~1/4th of patients achieve long-term remission without treatment; avoid indefinite therapy in non-responders23.
- Early discontinuation in non-responders reduces unnecessary treatment costs (~£113,623 saved per patient)23.
- Measuring response: Simple monitoring using patient feedback and 1–2 functional measures (e.g., grip strength, RODS score) is often sufficient to assess treatment response24.
- Biomarker data (e.g., neurofilament light chain) is inconsistent24; however, periaxin shows potential in distinguishing active vs. remission states25.
- For refractory patients, alternative options include CD20 depletion26,27,28,29(e.g., rituximab), mycophenolate, cyclophosphamide20, and emerging immunotherapies such as complement inhibitors30, BTKi31, ASCT32, and CAR-T therapy33.
Summary
- Shift from trial-and-error toward a mechanism-driven approach using early diagnostics and therapy response assessment.
- Pharmacokinetic-guided dosing and structured withdrawal trials can save healthcare costs and reduce patient burden.
- While current practice still depends on empirical decision-making, the future holds promise through biomarker validation and tailored immunotherapies to enable true personalization in CIDP care.
Abbreviations
ASCT: Autologous Stem Cell Transplantation, BTKi: Bruton's Tyrosine Kinase Inhibitor, CAR-T: Chimeric Antigen Receptor T cell, CIDP: Chronic Inflammatory Demyelinating Polyneuropathy, CD20: Cluster of Differentiation 20, MAG: Myelin-Associated Glycoprotein, FcRn: Neonatal Fc Receptor, IgG: Immunoglobulin G, IgM: Immunoglobulin M, IVIG: Intravenous Immunoglobulin, MRC: Medical Research Council, R-ODS: Rasch-built Overall Disability Scale, TTR: Transthyretin
References
- Caballero Avila M, et, al, Front Immunol 2025;16;1575464;2
- Hughes RAC, et al, Randomized Controlled Trial Lancet Neurol 2008,7(2):136-144
- Mendell JR et al, Neurology 2001,56(4):445-449
- Vermeulen M, et al, J Neurol Neurosurg Psychiatry 1993,56(1):36-39
- Thompson N, et al, Clinical Trial J Neurol 1996,243(3):280-285
- Hahn AF, et al, Brain 1996,119(4):1067-1077
- Hahn AF, et al, Brain 1996,119(4):1055-1066
- Mehndiratta MM, et al, Cochrane Database Syst Rev, 2015(8)CD003906
- Lieverloo GGA et al, J Neurol 2018,265(9):2052-2059
- Allen JA, et al, Lancet Neurol 2024,23(10):1013-1024
- Querol L, et al, Neurology, 2024,102(7_supplement_1)
- Fisto AL, et al, Neurol Res Pract, 2020,8:2:42
- Mahdi-Rogers M & Rajabally YA Biologics,2010,4:45-49
- Hadden RD, et al, Neurology 1999,53(1):57-61
- Nobile Orazio E, et al, Brain 2025,148(4):1112-1121
- Du Y, et al, Ann Clin Transl Neurol 2025,12(1):180-191
- Zheng Y, et al, J Peripher Nerve Syst 2024,29(3):350-355
- Allen JA and Lewis RA, Neurology 2015,85(6);498-504
- Breiner A et al, Muscle Nerve 2019,60(2);180-183
- Van den Bergh PYK, et, al, Eur J Neurol. 2021,28(11):3556-3583
- Cornblath, et al, Brain 2022 15;145(3):887–896
- Eftimov F. et al, Neurology, 2012,78(14):1079-1084
- Kapoor M. et al, J peripher nerv syst, 2021,26(4):461-468
- Rayabally YA et al, J Neurol Sci 2025; 744:123654
- Bellanti R and Lunn M. et al, submitted to Brain
- Hu J, et al, J Neurol. 2022;269(3):1250-1263
- Du Y, et al, Ann Clin Transl Neurol.2025;12(1):180-191
- Doneddu PE, et al, J Neurol Neurosurg Psychiatry,2024;95(9):838-844
- Nobile-Orazio, et al, Brain 2025;148(4):1112-1121
- Querol LA, et al, Neurotherapeutics 2022;19(3):864-873
- Rajabally YA, Immunotargets Ther 2024;13:99
- Axelson HW, et al, BMJ Case Rep. 2009ber092008.0927
- Zhang W, et al, hLife 2024;2(8):434-438
Agenda & Objectives
- To highlight disease burden, diagnostic challenges, and pathogenic mechanisms of CIDP, with a focus on:
- Clinical variability and diagnostic pitfalls
- Early axonal damage as a key driver of long-term disability
1) Disease Burden & Diagnostic Challenges
- CIDP is a chronic, immune-mediated disorder1 causing demyelination, axonal damage, and symptoms like proximal/distal weakness, sensory loss, and hyporeflexia2.
- Typical CIDP (proximal + distal weakness) is easiest to diagnose, while atypical variants (motor, sensory, distal, focal) often overlap with other conditions, complicating diagnosis3.
- Acute-onset CIDP-like presentations may indicate autoimmune nodopathies or Guillain-Barré syndrome4. Hematologic comorbidities (MGUS) may coexist but require distinct management5.
- Patients experience significant burdens like pain, muscle weakness, and frequent misdiagnoses6.
- Though CIDP is immune-mediated, early axonal damage is common7, predictive of long-term disability8, and highlights need for early recognition and prevention.
Summary
- A comprehensive understanding of CIDP’s clinical variability, early axonal involvement, and underlying complement-mediated mechanisms is essential to improving diagnostic precision, patient-centered care, and the development of targeted therapies that can reduce long-term disability.
2) Axonal Integrity and the Role of Complement
Objective
- To explore the physiological and pathological roles of the complement system in maintaining axonal integrity, and its relevance to CIDP and related neuromuscular diseases.
Complement System in CIDP Pathogenesis
- Axonal damage can occur early in CIDP7 and significantly contributes to cumulative disability8. Maintaining axonal integrity is a valid therapeutic approach8,9.
- Communication between neurons and Schwann cells supports nerve repair and function10. Mitochondrial function and turnover are essential due to high energy demand in neurons11.
- Beyond its cytotoxic or inflammation related functions, complement system helps maintain axonal integrity12.
- Classical (initiated by antibodies) and lectin (initiated by carbohydrates) pathways converge to activate C3 and C5, driving both protective and inflammatory responses13.
- Complement fragments (C3b, C3a, C5a) are involved in immune activation, opsonization, and inflammation14.
- Tight regulation by inhibitors such as CD59 is critical; dysfunction can lead to excessive tissue damage15.
- C1q and C3 mediate synaptic pruning during development, with C1q promoting axonal outgrowth, while downstream factors (C3) may inhibit it16.
- Complement fragments (C3a, C5a) play a role in tissue repair and promote inflammatory response to infection or injury17. Dysregulated complement activation contributes to neuroinflammation13.
- Complement-mediated pathology is implicated in diseases like CIDP, myasthenia gravis, etc17.
Summary
- The complement system has dual roles: maintaining axonal integrity (physiological) and driving pathology when dysregulated (e.g., in neuroinflammatory diseases).
- Targeting specific complement pathways (e.g., C1q, C3, MAC) offers therapeutic potential for PNS disorders.
- Approved complement inhibitors demonstrate clinical efficacy, reinforcing their relevance in neuromuscular disease treatment.
3) Demyelination and Axonal Damage Interplay in CIDP
Objective
- To examine the mechanisms contributing to demyelination and axonal injury in CIDP, and evaluate the role of complement activation, immune cell infiltration, and biomarkers in disease progression, diagnosis, and treatment monitoring..
CIDP pathophysiology & Complement’s Role in CIDP
- CIDP pathogenesis involves both cellular and humoral immune responses, creating a pro-inflammatory environment that damages both myelin and axons18.
- Complement activation (e.g., C3b, MAC formation) promotes demyelination and axonal damage18.
- Macrophages infiltrate nerves, targeting myelin sheaths (nodes of Ranvier, internodes) and small blood vessels18.
Axonal damage in CIDP
- Occurs early in disease progression (seen in nerve biopsies)19.
- Two proposed mechanisms (likely act in parallel, rather than exclusively):.
- Primary axonal injury: Antibodies bind paranodal proteins → complement activation → MAC formation → pore-induced axonal damage19.
- Secondary axonal injury: Demyelination → loss of axonal protection → MAC-mediated damage19.
- Axonal loss strongly correlates with long-term disability (more than demyelination alone)7.
Biomarkers
- Emerging biomarkers in CIDP include:
- Neurofilament light chain (NFL): associated with axonal damage and potential treatment monitoring (for axonal damage)20.
- Sphingomyelin: elevated in active CIDP (for myelin damage)21.
- C5a22 and IL-823: elevated in CIDP compared to controls; potential diagnostic utility (for inflammation).
- Peripherin: may help differentiate CIDP from other neuropathies like GBS (for axonal damage)20.
- MRI and ultrasound show promise for visualizing nerve enlargement and inflammation, and treatment response24.
Conclusion
- Demyelination and axonal damage in CIDP are interconnected, with complement activation (e.g., via MAC) contributing to injury. Early axonal involvement underscores the importance of timely treatment to prevent long-term disability.
- Although no single biomarker is definitive, a combined approach using fluid, imaging, and electrophysiological tools may improve diagnosis and treatment monitoring as evidence evolves.
4) Opportunities to target the pathobiology of CIDP
Objective
- To evaluate current and emerging treatment strategies for CIDP, focusing on mechanisms of action, therapeutic targets (e.g., complement, B-cells, FcRn), and future directions for personalized management to improve long-term outcomes.
Key highlights
- IVIG2, corticosteroids25, plasma exchange26, and FcRn antagonists26 are standard treatments with varied administration routes and onset times.
- Mechanisms range from immunoglobulin modulation and anti-inflammatory effects to antibody removal and immunosuppression27,28.
- Emerging Targeted Therapies:
- FcRn Antagonists: Reduce IgG levels by 70% by blocking recycling29, promoting lysosomal degradation30.
- B-Cell Depletion: Targets CD20+ B-cells (not plasma cells); limited evidence but used off-label28,31.
- Complement Inhibition: Upstream (C1q/C2) vs. terminal (MAC) blockade: Upstream inhibition may reduce inflammation and opsonization (vs. MAC-focused therapies like anti-C5). Potential to spare axonal damage if initiated early32.
- Personalized treatment approaches targeting specific immune mechanisms (FcRn, B-cell, complement) are a major area of interest.
- Biomarker development (fluid, imaging, and electrophysiologic) is essential for early diagnosis, treatment selection, and monitoring33.
- Addressing axonal damage in CIDP may prevent subclinical degeneration, reduce treatment side effects, minimize disability, and lower relapse and wear-off rates in incomplete responders34.
Conclusion
- A personalized, patient-centered approach that leverages biomarkers to guide early, targeted interventions (FcRn blockers, B-cell therapies, or complement inhibitors), offers the potential to improve outcomes, preserve axonal integrity, and reduce the overall treatment burden in CIDP.
Polling Results Highlighted by Speakers:
- Biggest challenge in CIDP: "Suboptimal response to current treatments in patients" and "timely accurate diagnosis" were top concerns.
- Biomarker utility: Majority voted for "treatment monitoring" (NFL favored for axonal damage).
- Future research priority: "Personalized treatment approaches" topped the poll.
Abbreviations
C1/2/3/5: Complement component 1/2/3/5, CD20/59: Cluster of Differentiation 20/59, CIDP: Chronic Inflammatory Demyelinating Polyneuropathy, FcRn: Neonatal Fc Receptor, GBS: Guillain-Barré Syndrome, IgG: Immunoglobulin G, IL-8: Interleukin 8, IVIG: Intravenous Immunoglobulin, MAC: Membrane Attack Complex, MGUS: Monoclonal Gammopathy of Undetermined Significance, MRI: Magnetic Resonance Imaging, NFL: Neurofilament light chain, PNS: Peripheral Nerve Society
References
- Broers MC, et al. Neuroepidemiology. 2019;52(3-4):161–172
- Bunschoten C, et al. Lancet Neurol. 2019;18(8):784-794
- Allen J. Neurol Ther. 2020;9:43-54
- Lewis RA, et al. J Neurol Sci. 2022;443:120478
- Mathey EK, et al. J Neurol Neurosurg Psychiatry. 2015;86(9):973-985
- https://www.gbs-cidp.org/support/connect-with-gbs-cidp-community/patient-stories/
- Al-Zuhairy A, et al. Clinical Neurophys. 2021;132(4):1000-1007
- Grüter T, et al. Eur J Neurol. 2022;29(2):583-592
- Ricciardi D, et al. Brain Sci. 2022;12(11):1510
- Trends Neurosci. 2012;35(2):123-134 (https://pubmed.ncbi.nlm.nih.gov/22192173/)
- Chamberlain KA, Sheng ZH. J Neurosci Res. 2019;97(8):897-913
- Yuan Y, et al. Front Neurol. 2022;13:908148
- Schartz ND, Tenner AJ. J Neuroinflammation. 2020;17(1):354
- Liszewski MK, Atkinson JP. Hum Genomics. 2015;10;9(1):7
- Karbian N, et al. J Neuroinflammation. 2023;20(1):245
- Warwick CA, et al. J Biol Chem. 2021;297(3):101085
- Dalakas MC, et al. Nat Rev Neurol. 2020;16(11):601-617
- Querol LA, et al. Neurotherapeutics. 2022;19(3):864-873
- Al-Zuhairy A, et al. Muscle Nerve. 2022;66(6):715-722
- Keddie S, et al. Brain. 2023;146(11):4562-4573
- Capodivento G, et al. J Neurol Neurosurg Psychiatry. 2021;92(3):303-310
- Quast I, et al. Ann Clin Transl Neurol. 2016;3(9):730-735
- Kmezic I, et al. Front Immunol. 2023;14:1241199
- Härtig F, et al. Neurother. 2018;15(2):439-451
- Hughes RAC, et al. Cochrane Database Syst Rev. 2017;11(11):CD002062
- Mina-Osorio P, et al. Transfus Med Rev. 2024;38(1):150767
- Mair D, et al. J Neurol Neurosurg Psychiatry. 2025;96:38-46
- Briani C, Visentin A. Neurotherapeutics. 2022;19:874-884
- Mina-Osorio P, et al. Transfus Med Rev. 2024;38(1):150767
- Gable KL, Guptill JT. Front Immunol. 2020;10:3052
- Forsthuber TG, et al. Ther Adv Neurol Disord. 2018;11:1756286418761697
- Querol L, et al. J Peripher Nerv Syst. 2023; 28(2):276-285
- Dalakas MC. Nat Rev Neurol. 2011;7(9):507-517
- Ryan M, et al. AJMC. 2018;24(17):S371–S379
Agenda & Objectives
- To deepen the understanding MMN immunopathogenesis and the critical role of complement activation in its progression
- To raise awareness on the importance of monitoring disease activity in MMN management, focusing on using MMN-RODS as a key tool
- To highlight the needs and expectations of patients with MMN regarding future therapeutic approaches
Patient’s perspective – Living with MMN
At 44, a male patient began with right shoulder pain, misdiagnosed as a sports injury. Symptoms worsened to muscle atrophy, drop wrist, and severe weakness, impacting daily tasks. After multiple inconclusive tests and a misdiagnosis of ALS, he was finally diagnosed with MMN after seeing seven neurologists and 25 healthcare professionals.
- This case stresses the need for quantitative tools in clinical discussions as current monitoring is mostly qualitative. The median diagnosis time for MMN can be up to five years, and untreated disease severity worsens.
MMN pathogenesis and the role of complement
- MMN is an autoimmune disorder driven by IgM anti-GM1 antibodies, which activate the classical complement pathway1.
- These antibodies bind particularly at the node of Ranvier—a site uniquely lacking in complement regulatory proteins like CD59—making it especially vulnerable to membrane attack complex (MAC)-mediated damage2,3.
- Complement activation not only damages membranes but also triggers an inflammatory cascade involving cytokines, chemokines, and antigen presentation1,4.
- This multi-step process includes antibody leakage, nerve binding, reversible conduction block, and ultimately axonal degeneration and muscle atrophy, explaining the patient’s sudden losses in strength1,4.
- Therapeutically, complement pathway inhibition offers promise. Depending on the target—such as C1 in the classical pathway or C5 for terminal MAC formation—different parts of the cascade can be modulated, each with different clinical implications5,6.
- Understanding this immunopathogenesis is essential for both developing therapies and interpreting patient responses1,7.
Measuring MMN Outcomes: The MMN-RODS
- Historically, a variety of metrics have been applied such as Grip strength10 (Martin Vigorimeter), MRC scores11, 9-Hole Peg Test12, GBS Disability Score, EQ-5D, etc. to measure MMN progression and treatment efficacy. However, these vary in responsiveness8, reliability9, and cross-cultural validity13,14.
- The MMN-RODS (Rasch-built Overall Disability Scale) was developed to address these gaps. Initially distilled from 146 daily activities, it now includes 25 items focusing on fine motor skills, capturing daily function with three simple response options.8 Though it appears simple, the scale transforms responses into linear measures suitable for statistical analysis and parametric modeling8.
- MMN-RODS has been validated in multiple studies, demonstrating high internal consistency, convergent validity, and promising responsiveness.15 It is currently being evaluated further in ongoing phase 2 and 3 trials involving over 50 patients across 40 sites in Europe and North America.16 The goal is to strengthen its utility in both clinical practice and research.15
Abbreviations
ALS: Amyotrophic Lateral Sclerosis, CD59: Cluster of Differentiation 59, CIDP: Chronic Inflammatory Demyelinating Polyneuropathy, C1: Complement component 1, C5: Complement component 5, EQ-5D: EuroQol 5 Dimensions, GBS: Guillain-Barré Syndrome, GM1: Ganglioside M1, IgM: Immunoglobulin M, MAC: Membrane Attack Complex, MMN: Multifocal Motor Neuropathy, MMN-RODS: Multifocal Motor Neuropathy Rasch-built Overall Disability Scale, MRC: Medical Research Council
References
- Harschnitz O, et al. J clin Immunol. 2014;34(suppl 1):S112-S119
- Vlam L, et al. Nat Rev Neurol. 2011;8(48):48-58
- Karbian N, et al. J Neuroinflamm. 2023;20(1):245
- McGonigal R, et al. J Clin. Invest. 2022;132(14):e158524
- Garred P, et al. Pharmacol Rev. 2021;73(2):792–827
- Ricklin D, et al. Nat Rev Drug Discov. 2019
- Beadon K, et al. Curr Opin Neurol. 2018;31(5):559-564
- Vanhoutte EK, et al. J Peripher Nerv Syst. 2015;20(3):296-305
- Pruppers MHJ, et al. J Peripher Nerv Syst. 2015;20:306-318
- Neumann S, et al. In Vivo. 2017;31(5):917-924
- Chia PK, et al. J Clin Neurosci. 2019;69:114–119
- Moreno-Morente G, et al. Int J Environ Res Public Health. 2022;19(16):10080
- Van Nes SI, et al. J Peripher Nerv Syst. 2009;14:268–278
- Sharrack B and Hughes RAC, Multi Scler. 1999;5(4):223-233
- ArgenX. Data on File. 2025 (Data on File will be made available from ArgenX upon request)
- https://clinicaltrials.gov/ NCT05225675
Expert Video Interviews
Dr. Tina Dysgaard (Denmark)
Dr. Dysgaard shares her insights on the impact of subcutaneous immunoglobulin (SCIG) on CIDP management, discussing the challenges of transitioning patients to SCIG and the criteria for patient eligibility. She also explores the role of SCIG in different countries and its potential benefits.
How SCIG has influenced CIDP management
SCIG therapy initiation
Challenges when switching to SCIG
Role of SCIG in other countries
Prof. Marie Theaudin (Switzerland)
Prof. Theaudin discusses the management of CIDP at her center, emphasizing the criteria for patient selection and sharing a detailed patient case. Her insights highlight the importance of personalized care and the challenges faced in clinical practice.
Management of CIDP in the Centre hospitalier universitaire vaudois (CHUV)1, 2
Criteria for patient selection
A patient case
Dr. Paolo Ripellino (Switzerland)
Dr. Ripellino provides an overview of his congress highlights, focusing on the integration of new therapies into clinical practice. He also discusses the criteria for determining patient eligibility for home treatment, offering valuable perspectives on advancing CIDP management.
Congress highlights3
Integrating new therapies
Criteria for home treatment
Abbreviations:
PNS, Peripheral Nerve Society; SCIG, Subcutaneous Immunoglobulin; CIDP, Chronic Inflammatory Demyelinating Polyneuropathy
References:
Van den Bergh, Peter Y K et al. “European Academy of Neurology/Peripheral Nerve Society guideline on diagnosis and treatment of chronic inflammatory demyelinating polyradiculoneuropathy: Report of a joint Task Force-Second revision.” Journal of the peripheral nervous system : JPNS vol. 26,3 (2021): 242-268.
Kuwabara, Satoshi et al. “Intravenous immunoglobulin for maintenance treatment of chronic inflammatory demyelinating polyneuropathy: a multicentre, open-label, 52-week phase III trial.” Journal of neurology, neurosurgery, and psychiatry vol. 88,10 (2017): 832-838.
Allen, Jeffrey A et al. “Safety, tolerability, and efficacy of subcutaneous efgartigimod in patients with chronic inflammatory demyelinating polyradiculoneuropathy (ADHERE): a multicentre, randomised-withdrawal, double-blind, placebo-controlled, phase 2 trial.” The Lancet. Neurology vol. 23,10 (2024): 1013-1024.
VV‑MEDMAT‑133174

